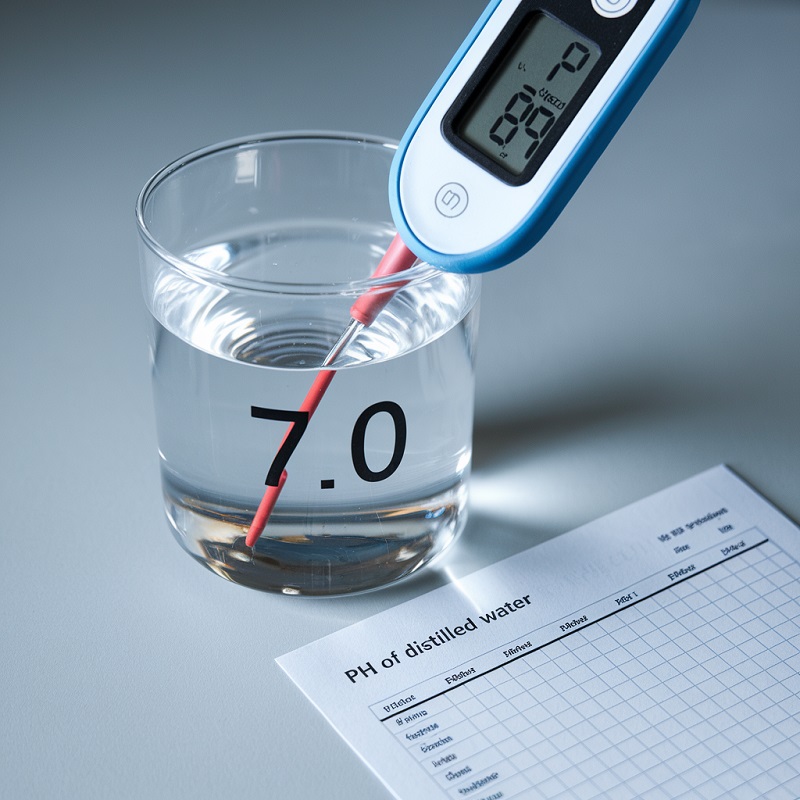
The pH of distilled water refers to its stage of acidity or alkalinity on a scale from 0 to 14. Pure distilled water has a pH of seven, which is taken into account impartial. Nevertheless, this will change below sure circumstances.
What’s pH? Why is it an Necessary Issue for Water and Different Substances?
pH stands for “potential of hydrogen” and measures the focus of hydrogen ions in an answer. It determines whether or not a substance is acidic, impartial, or fundamental:
- Acidic (pH < 7): Substances with extra hydrogen ions, like lemon juice or vinegar.
- Impartial (pH = 7): Pure water, neither acidic nor fundamental.
- Primary (pH > 7): Substances with fewer hydrogen ions, like baking soda or bleach.
Significance of pH in Distilled Water
- Water High quality: The pH of water impacts its style, security, and the way effectively it helps aquatic life.
- Well being & Medication: Blood pH should stay steady (round 7.4) for correct physique operate.
- Agriculture: Soil pH impacts plant progress and nutrient availability.
- Industrial Processes: Many industries require particular pH ranges for manufacturing.
What Does a Excessive pH Imply?
- Excessive pH (Above 7): Signifies alkalinity. Water with a excessive pH could style bitter and kind scale deposits in pipes and home equipment.
- Instance: Alkaline water, usually marketed for well being advantages, usually has a pH between 8 and 9.
What Are the Dangers of Consuming Unbalanced pH Water?
Ingesting water with an unbalanced pH can pose a number of well being and sensible dangers:
- Well being Dangers:
- Acidic Water (pH < 6.5):
- Tooth Decay: Acidic water can erode tooth enamel.
- Digestive Points: It might trigger irritation within the abdomen and intestines.
- Metallic Contamination: Acidic water can corrode pipes, leaching metals like lead and copper into consuming water.
- Alkaline Water (pH > 8.5):
- Digestive Issues: Can disrupt pure abdomen acid, resulting in digestion points.
- Mineral Imbalance: Extreme alkalinity could cut back the physique’s potential to soak up important minerals.
- Kidney Stress: Folks with kidney circumstances ought to keep away from high-alkaline water.
- Technical and Family Dangers:
- Plumbing Harm: Water with a really low or excessive pH can injury plumbing by corroding pipes or inflicting mineral buildup.
- Equipment Put on: Dishwashers, washing machines, and occasional makers could endure from scale buildup or corrosion.
- Aquatic Life Hurt: In aquariums, improper pH can hurt fish and different aquatic organisms.
Actual-World Instance:
Why is pH Necessary?
The pH scale measures how acidic or fundamental a substance is. Right here’s why understanding the pH of distilled water issues:
- Well being Functions: Distilled water is utilized in laboratories and hospitals as a result of it is freed from impurities and protected for medical use.
- Scientific Analysis: Scientists depend on distilled water as a impartial baseline in experiments, guaranteeing correct and unbiased outcomes.
- Residence Makes use of: Folks use distilled water for family duties equivalent to watering crops and filling aquariums to keep away from mineral buildup and keep a steady setting.
How is Distilled Water Made?
Distillation includes boiling water, capturing the steam, and condensing it again right into a liquid. This course of removes impurities, together with minerals and salts. Learn Extra : Water Distiller Price It?
Comparability of Water Sorts by Purity
| Water Kind | Course of | pH Vary |
| Distilled Water | Boiled & Condensed | 6.5 – 7.0 |
| Faucet Water | Municipal Provide | 6.0 – 8.5 |
| Spring Water | Pure Supply | 7.0 – 8.5 |
| Mineral Water | Mineral Enriched | 7.0 – 8.5 |
Why is Distilled Water Not At all times pH 7?
Whereas distilled water is initially impartial, a number of elements can alter its pH:
- Publicity to Air: Distilled water absorbs carbon dioxide from the air, forming carbonic acid, which lowers its pH.
- Storage Container: Plastic containers could leach chemical compounds into the water, altering its pH.
- Storage Length: The longer distilled water is saved, the extra its pH can change as a result of environmental publicity.
pH of Distilled Water vs. Faucet Water
Distilled water is impartial, whereas faucet water’s pH can differ based mostly on its supply and therapy.
Instance:
- Distilled Water Pattern (Lab Check): pH 7.0
- Faucet Water from New York: pH 7.2 (handled with chlorine to disinfect)
- Faucet Water from California: pH 8.1 (accommodates pure minerals that improve alkalinity)
Functions of Distilled Water by pH
- Medical Use: Distilled water is clear and protected for medical procedures, equivalent to sterilization and wound cleansing.
- Scientific Experiments: Its impartial pH ensures correct measurements and reactions in laboratory experiments.
- Automobile Batteries & Home equipment: It prevents mineral buildup in delicate gadgets like automotive batteries and humidifiers.
- Pores and skin Care & Cosmetics: Attributable to its purity, distilled water is mild on the pores and skin and is commonly utilized in beauty merchandise.
How you can Measure the pH of Distilled Water
You may measure pH utilizing:
- pH Strips: Easy and reasonably priced, these strips change colour based mostly on the water’s pH stage.
- Digital pH Meter: A extra exact software that shows the precise pH worth on a digital display.
- Chemical Check Kits: Frequent in laboratories, these kits provide extremely correct readings by way of chemical reactions.
By understanding the pH of distilled water, you may make higher choices about its makes use of in on a regular basis life, science, and health-related duties.